
Where is the water going?
Anthropogenic climate change is warming the planet. At the time of this writing there is 420 ppm of CO2 in the atmosphere and air temperatures are on the order of 1.5 °C above the preindustrial period. Without significant emissions reductions, we are facing 2 °C of warming by 2050.
The water systems of the western USA, and many other parts of the world, rely on the fact that water falls in the ice-phase (as snow) and sits around on the ground for a while, melting slowly.
So there are many open questions about what happens to snow, snowmelt, and runoff into rivers as snow climates continue to warm.
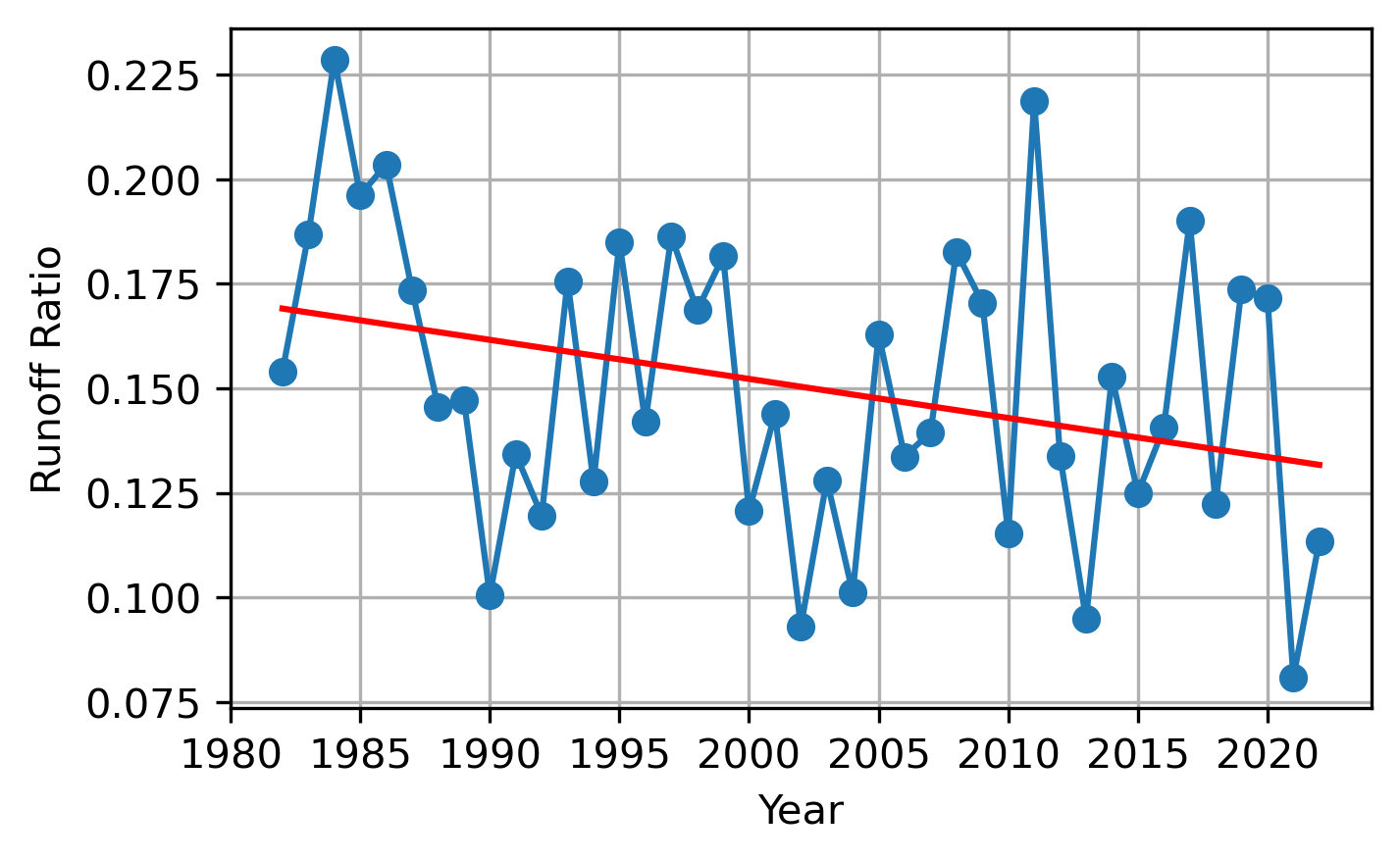
Among these are whether the runoff ratio – defined as the amount of runoff relative to precipitation (rain and snowfall) – declines as the snow climate warms. I discussed this in a previous post. The figure below shows possible evidence of declining runoff ratios in the Upper Colorado river basin. (Note that, in this very arid basin, that the runoff ratio is very low to begin with! Meaning that most precipitation leaves the land by evaporation and/or change in groundwater storage).
In snow-dominated regions, it is commonly suggested that sublimation increases with rising air temperatures, meaning that more water leaves the snowpack into the atmosphere rather than melting and entering rivers and reservoirs. Yet sublimation remains a very complex topic, particularly in real-world conditions. I’m going to walk through some arguments that suggest sublimation will probably be less important in the future, not more and that other evaporative mechanisms may increase at a faster rate as temperatures rise.
Sublimation science basics
Thinking about sublimation means thinking about two coupled processes: energy and mass (water vapor).
Energy
Sublimation is the phase change of ice to water vapor. It takes 2.835E6 joules to completely sublimate one kilogram of ice. On the other hand, it takes 2.501E6 joules to evaporate one kilogram of water from the liquid phase to the vapor phase. Thus, from an energy perspective, it takes about 12% more energy to sublimate ice than to evaporate liquid water. You can maybe already see where my argument is going.
Mass exchange
The energy budget is not the only consideration in this coupled system. Vapor exchange between the snow and the atmosphere is modelled as a turbulent diffusion process.
The bulk formula for evaporation may be expressed as:
\[E \approx \mathrm{C}_D \cdot wind \cdot \Delta humidity\]Where \(E\) is evaporation (or sublimation), C_D is a coefficient between 0 and 1 that describes the effects of turbulence, and \(\Delta humidity\) is the difference between the humidity in the air and the humidity of the surface.
One convenient way of measuring vapor is using the partial atmospheric pressure of water vapor (e.g., vapor pressure) which can be converted to mass-units. The vapor pressure of ice is determined by using the saturation vapor pressure formula.
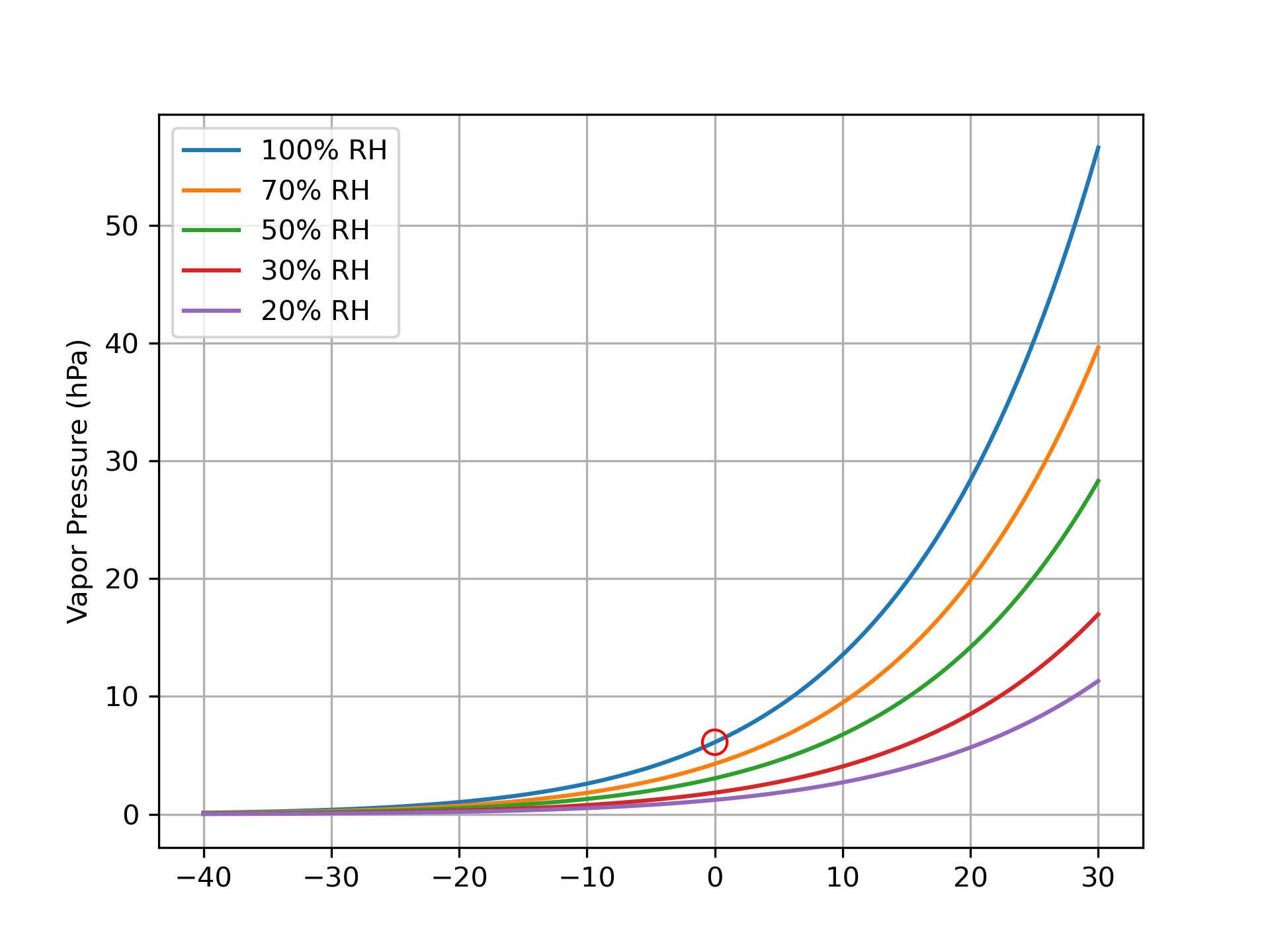
For water vapor, this is given by the Clausius-Clapeyron relationship. There are slight modifications with respect to ice, which state that the saturation vapor pressure of ice is slightly less than that of an equivalent liquid surface 1. This is a very non-linear relationship, meaning that the saturation vapor pressure rapidly ramps up with warmer temperatures. This is another way that the mass and energy fluxes are coupled, since the surface humidity depends on the temperature.
And here’s a fun fact about snow: it never gets above 0 °C! If it does, it melts and becomes water. Therefore the vapor pressure of the snow surface is fundamentally capped at 6.11 hPa. It can never exceed this value. Vapor pressure of water in the atmosphere – or liquid water on the earth’s surface at typical temperatures — does not have the same thermodynamic limit.
So when does snow sublimate? This happens when the vapor pressure of the snow exceeds the vapor pressure of the air. In the absence of fog or clouds, the air (unlike the snow) will not be completely saturated (e.g., RH=100%) with respect to its temperature. The RH of the air can drop quite low.
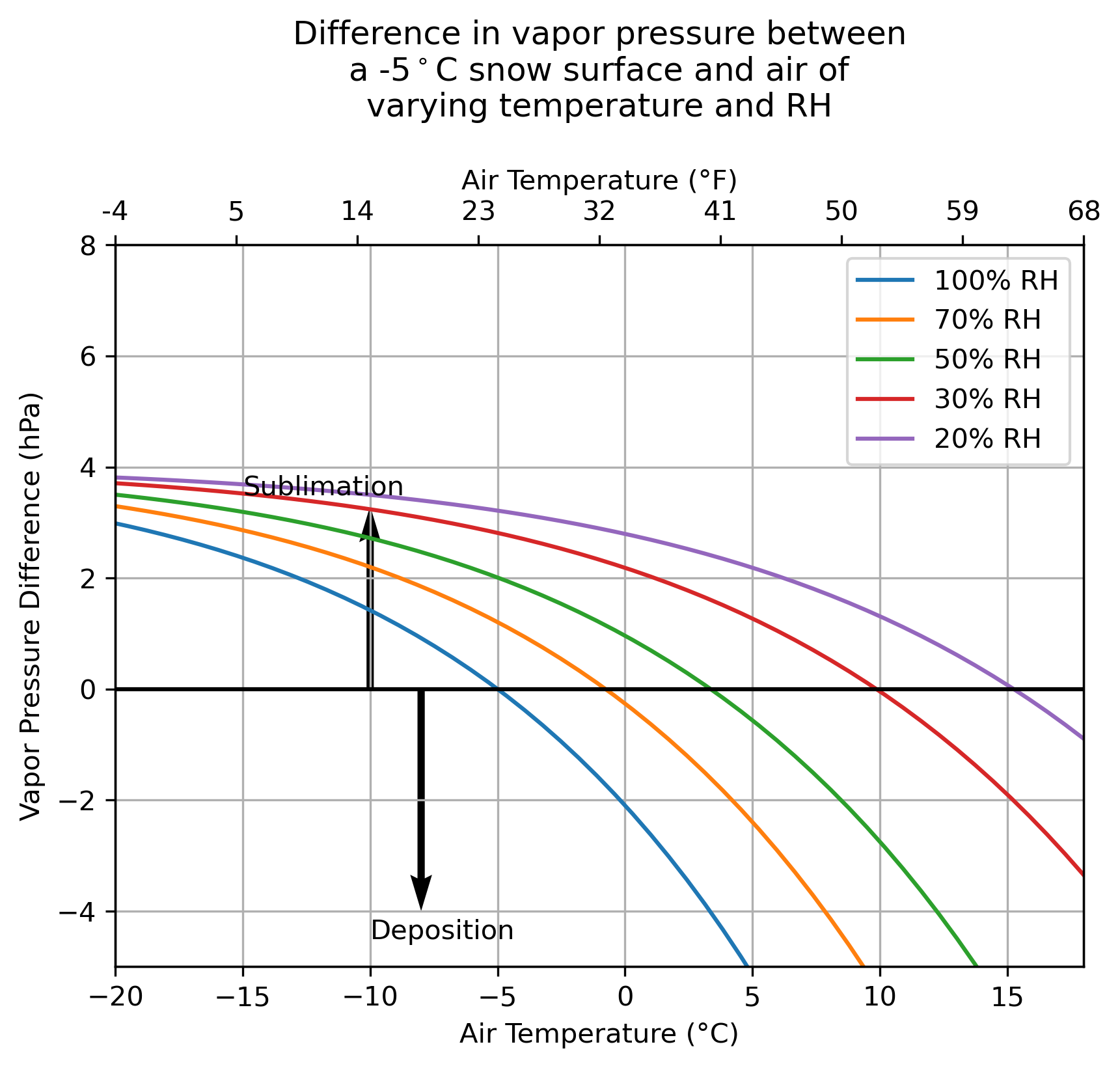
We can look at the hypothetical air-snow vapor pressure difference between air of various relative humidities and temperatures and snow at a constant temperature. We can start with a snow surface at -5 °C.
And then look at a snow surface where the maximum temperature is 0 °C (which is again the maximum vapor pressure of snow.)
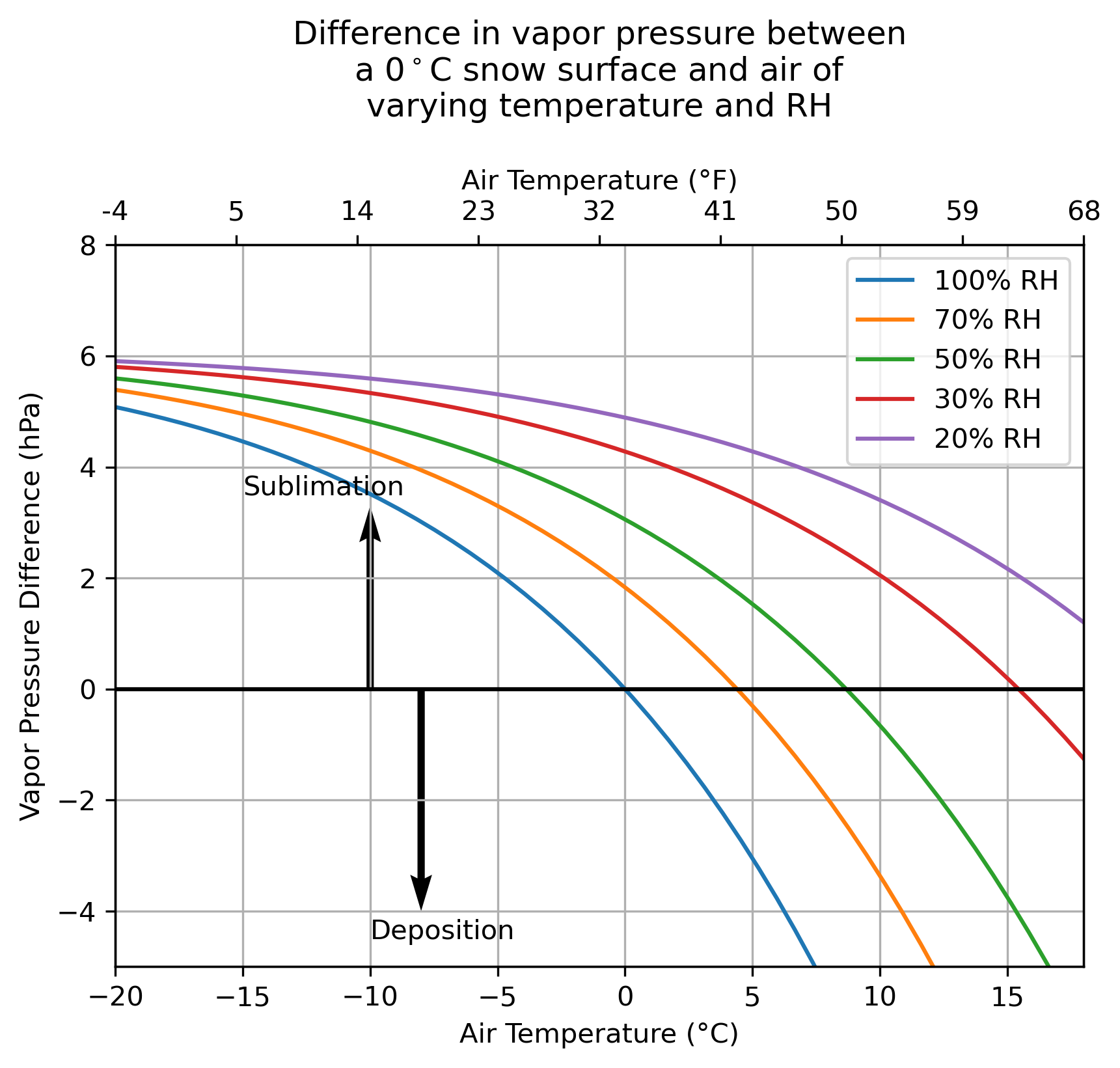
I think the natural intuition is that we should get more and more sublimation as it gets warmer — but we find that this is not necessarily the case! At a constant RH, warmer temperatures actually weaken the vapor pressure gradient between the snow and the air, weakening sublimation.
Also, at 0 °C the snow is probably melting, not sublimating. It has been shown that both processes probably co-occur, but this is an area that merits additional investigation.
Example Case: The March 2026 heatwave at CSSL
Looking at some data for the Central Sierra Snow lab (CSSL) in Soda Springs, CA shows the RH and temperature values that occurred during the March 2026 heatwave.
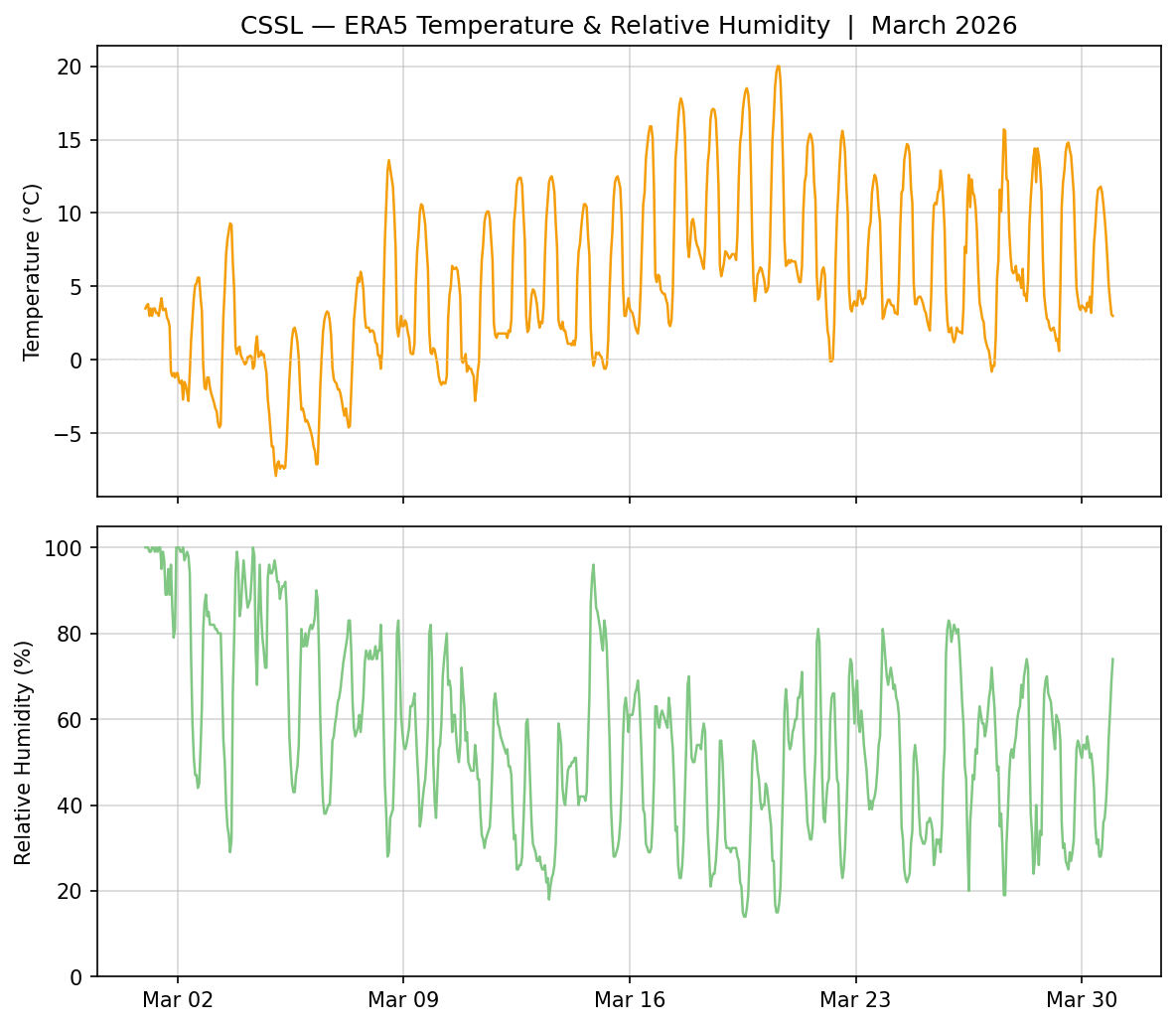
We see that relative humidity 1) has a diurnal pattern and is lowest during the daytime, and 2) reaches low values of less than 20% during the hottest part of the heatwave. Combining this data with the previous charts, we can see that a very low RH of 20% and an air temperature of 15 °C does not actually lead to a significant vapor pressure gradient over a -5°C snowpack.
However, during this event, the actual snow temperature was probably closer to 0 °C (since the snow was consistently melting; see the previous post). This would lead to a ~2 hPa gradient, which is about the same as an airmass near freezing with an RH of 70%. This may be a counterintuitive result, but the point is that both the humidity and temperature of the air are important, as well as the snow temperature itself. Warmer temperatures do not necessarily imply a stronger and stronger rate of sublimation based on vapor pressure arguments alone.
Of course there is another way of looking at this that could suggest sublimation may increase climatologically. If, on average, a snowpack spends more time closer to 0 °C than -5 °C, then we can see that the sublimation gradient increases in magnitude for a given air temperature, which would also imply some snow-climate dependency on the sensitivity of sublimation to warming.
The case against sublimation
Bare-ground evaporation
Once the snowpack has melted, the process of converting water stored on the land to water-in-the air continues at a rapid clip. There are two important considerations for thinking about evapotranspiration. As the name implies, water travels through both evaporation from the bare-soil or pooling liquid water and an active biological process: “transpiration”. Let’s think about evaporation from bare-ground first.
The following plot shows data from the SAIL field campaign in Crested Butte, CO. These instruments measure the air temperature, the temperature of the snow and/or ground surface, and also the albedo of the land using up/down looking pyranometers. We can see a fascinating effect. As soon as the snow melts (as indicated by the albedo going from ~0.80 or 0.90 to 0.18) we see that the surface temperature absolutely rockets upwards! It exceeds 35 °C midday in many cases. Notice that, when the snowpack is present, the retrieved surface temperature seldom exceeds 0 °C. This happens for several reasons, including that the ground starts absorbing much more solar radiation than the snow.
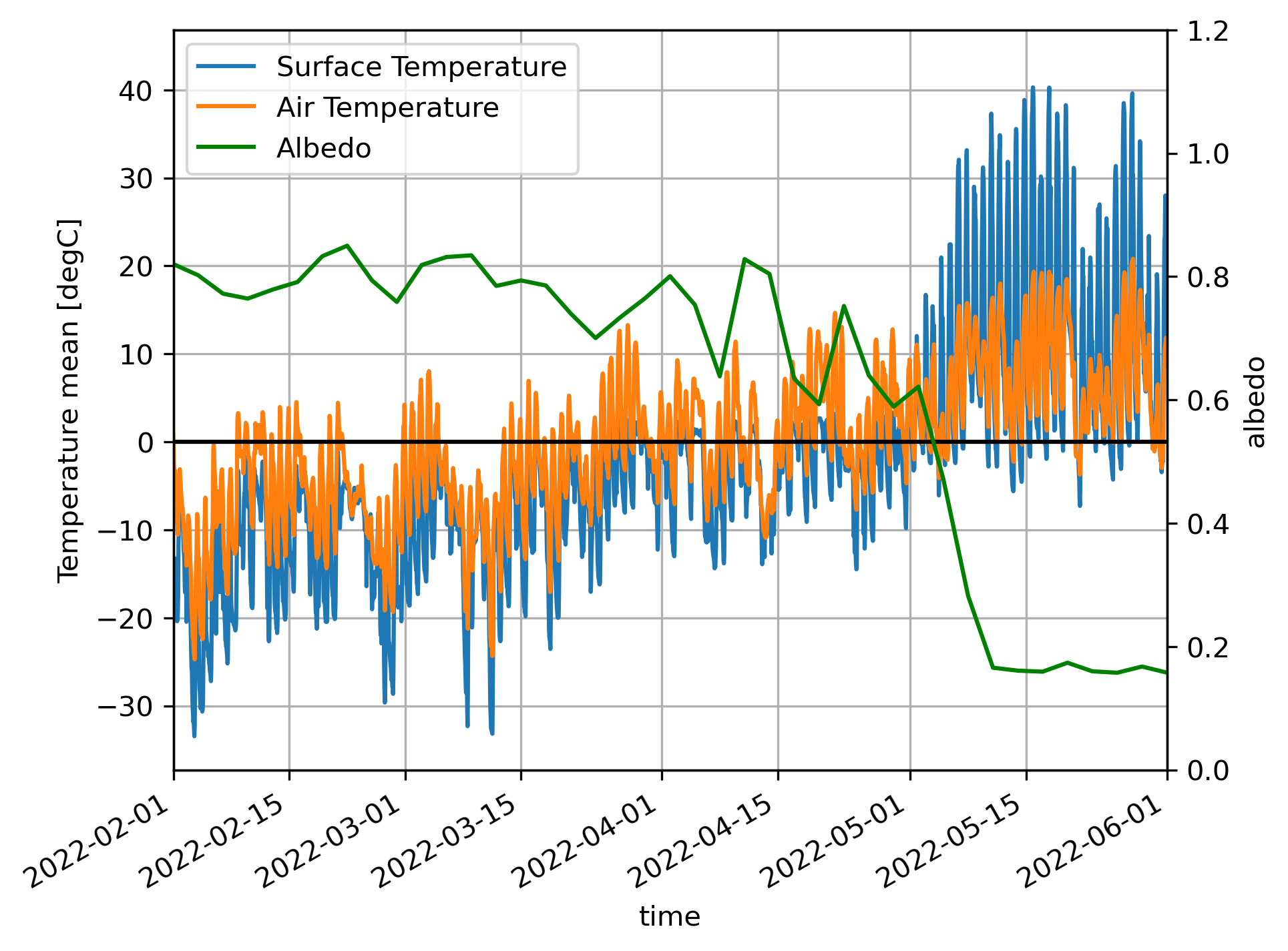
What does the transition from snow to bare-ground mean for sublimation/evaporation?
- For one, converting liquid soil-water to vapor requires less energy than ice to vapor.
- Second, there’s more energy available now because of the reduced albedo.
- Third, there is no longer a thermodynamic cap on the surface vapor pressure — it can and does exceed 6.11 hPa quite easily!
- Fourth, snow is very smooth with a low aerodynamic roughness compared to bare ground or short vegetation. This impacts the “Cd” parameter in the evaporation equation. All else being equal, a smaller roughness will have a lower Cd.
- Fifth, the loss of snow means we have transitioned from a mostly stable surface layer to an unstable surface layer. When air is warmer than the snow the surface layer is “stable” (I discuss this in the previous post) meaning that turbulence is suppressed. When the surface is warmer than the air, as it often is once snow melts, we enter an unstable regime — this is great for promoting turbulent eddies and enhancing evaporation. Stable layers will have a lower Cd value and unstable layers will have a higher Cd value.
And let’s go back to the vapor pressure argument. How does the vapor pressure difference (between a hypothetical surface be it snow or saturated soil) scale as a function of surface temperature? We can imagine different air-to-surface temperature differences (denoted by \(\Delta T\)) and an atmosphere with a constant 30% RH.
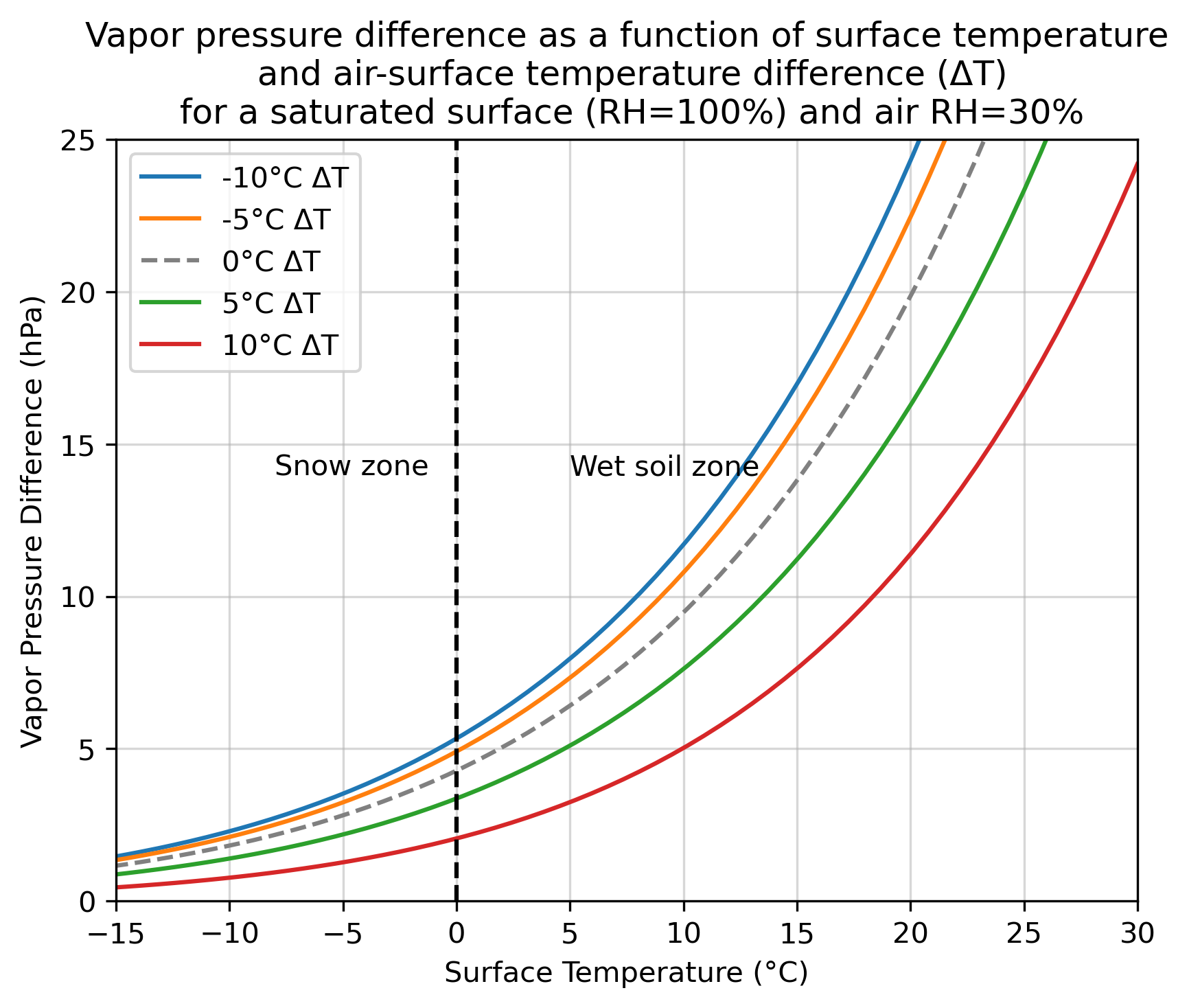
(To help interpret the chart above: a \(\Delta T\) of 5 °C means the air is 5 °C warmer than the surface, which is given by the value on the x-axis. So, a surface temperature of 10 °C would mean the air temperature is 15 °C in that case).
So, we can see that for a range of plausible values, that the vapor pressure difference explodes as the surface warms. Not all the plausible values are even within the y-axis of the plot.
Transpiration
After snow melts, water may pool on the surface and evaporate, but also diffuse into lower levels of the soil column. Does that mean it’s safe from leaving into the atmosphere? Not at all. Like us, trees also like to drink water. Whereas bare soil evaporation primarily happens relatively close to the surface, plants have roots that draw soil moisture from deep within the ground. A complete theoretical unification of evaporation theory that encapsulates idealized snow or bare-ground surfaces with transpiration through the stomata of leaves is still not completely unified, though models nonetheless work towards capturing these effects.
A key starting point for thinking about transpiration is the Leaf-area index, which expresses the areal extent of leaf area per areal extent of the land surface. The numbers are generally above unity — e.g., leaves occupy much more surface area than bare ground, and are therefore great at exchanging water vapor/CO2/O2 with the atmosphere. A full science argument about the effects of rising temperature — and direct CO2 fertilization effects – on water use is beyond the scope of this article and suffice to say an area of active research. But the point is that transpiration should not be discounted as a major factor in the water budget of high elevation, snow dominated regions.
Conclusions

This is far from a complete scientific argument about the relative importance of sublimation versus other evaporative processes as a function of warming. The arguments against the importance of sublimation mostly stem from the 0 °C limitation — snow cannot exceed a 6.11 hPa vapor pressure, and warm air temperatures over a cold-surface inhibit turbulence (stability feedback). From the energy perspective, the argument against sublimation is that snow has a high albedo – net radiation increases rapidly as soon as snow melts, so there is much more available energy to evaporate soil-water rather than sublimate snow. Finally, it takes about 12% less energy to convert water to vapor than ice to vapor.
The most compelling argument in favor of sublimation may look something like this. Cold snowpacks with average snow temperatures well below freezing, will in principle, become more likely to sublimate as they warm purely because of the non-linear vapor pressure effect. Snowpacks sit around on the landscape for a long time, leaving ample time exposed to the atmosphere and therefore a chance to sublimate. Liquid water, on the other hand, flows under the force of gravity and drains into the soil and streams where it may not have the chance to evaporate or transpire.
There are many interacting components at play — including feedbacks from the land to atmospheric humidity and temperature that are not discussed here— and a complete accounting requires careful model analysis and field observation. This is far from a closed case, but in this post I have laid out a first-principles argument that factors other than sublimation are likely more important for understanding declining runoff ratios.
-
Climatically this is actually an incredibly important process — at low pressures higher up in the atmosphere, the lower saturation vapor pressure of ice relative to liquid water is nearly a requirement for snowfall to grow (the Bergeron-Findeisen process). There are also curvature effects (Kelvin effect) that matter at small crystal sizes. Clausius-Clapeyron and related formulae are strictly defined for an infinite flat surface. ↩